OPEN-ACCESS PEER-REVIEWED
REVIEW ARTICLE
Natasa P. Kalogiouri and Victoria F. Samanidou*
Laboratory of Analytical Chemistry, Department of Chemistry, Aristotle University of Thessaloniki, Thessaloniki GR 54124, Greece.
Reviews in Separation Sciences. Vol.1. No.1. pages 17-33 (2019).
Published 15 October 2019. https://doi.org/10.17145/rss.19.003 | (ISSN 2589-1677).
*Correspondence:
Samanidou VF. . Laboratory of Analytical Chemistry, Department of Chemistry, Aristotle University of Thessaloniki, Thessaloniki GR 54124, Greece.
Editor: Dr. Mohammad Sharif Khan, Thayer School of Engineering, Dartmouth College, Hanover NH, USA.
Open-access and Copyright:
©2019 Kalogiouri N and Samanidou VF. This article is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) which permits any use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Funding/Manuscript writing assistance:
The authors have no financial support or funding to report and they declare that no writing assistance was utilized in the production of this article.
Competing interest:
The authors have declared that no competing interest exist.
Article history:
Received 7 June 2019, Revised 29 July 2019, Accepted 30 July 2019.
Abstract
Antioxidants represent a class of naturally occurring molecules that are responsible for several health properties. Antioxidants are present in various natural products and food matrices, typically known as “functional foods”. Because of their high nutritional value and antioxidant activity, functional foods gain considerable attention, in recent years. Phenolic compounds are the most abundant class of antioxidants and Liquid Chromatography (LC) is the main technique used for their separation. Conventional High-Pressure Liquid Chromatography provides insufficient resolving power and requires long analysis times. New techniques like ultra-high-pressure liquid chromatography (UHPLC), in reversed-phase (RP) or hydrophilic interaction (HILIC) mode, and the development of multidimensional methods prove promising with increasing applications for phenolic analysis. The aim of this review is to provide an updated overview of the applications of recent advances in LC phenolic separation of antioxidants in functional within the period 2015-2019.
Keywords
Functional Foods, Separation, Antioxidants, Liquid Chromatography, Analytical Techniques.
1.0 Introduction
Current trends in the food industry focus on the production of innovative functional foods, which possess a significant place in modern diet [1]. Still, there is not an official definition for functional foods, but under convention the term describes foods that have been designed in such a way by the industry, with the contribution of chemistry and nutrition science, to provide health benefits [2,3]. The main objective is, of course, to create innovative products that are distinguished for their appearance, texture and special organoleptic characteristics in order to attract consumers’ interest. There are two categories of functional foods, those that are physically functional and those with added value, formulated from natural constituents in order to target certain physical and biological functions [4]. Food constituents with functional properties are studied and extracted in order to be used as physical additives, providing both advanced technological properties and health benefits [5,6].
Among physically functional, plant-based foods such as seeds, olive drupes and olive oil, honey, nuts, grapes, and berries are rich in antioxidants and have supported clinical health claims [7,8]. Between natural antioxidants, phenolic antioxidants gain particular interest since these compounds inhibit free radical formation and/or interrupt propagation of autoxidation [9]. At laboratory scale, different separation techniques have been developed for the determination of phenolic antioxidants from food matrices, commonly known as “bioactive phenolic compounds”. The general term “bioactive” describes the naturally occurring physical constituents of foods presenting antioxidant activity and have the capacity to modulate metabolic processes demonstrating beneficial effects, such as induction and inhibition of enzymes and gene expression, providing protection against many diseases, including cancer [10-12]. Bioactive compounds vary in function and chemical structure and thus, are grouped accordingly. Among bioactive compounds, polyphenols and simple phenols are the most abundant antioxidants and gain particular attention, since they have been associated with numerous nutritional and functional properties [13,14]. They contribute to the organoleptic properties, such as aroma, taste, and color, showing antioxidant activity, as well [15].
The analysis of phenolic antioxidants remains challenging because of their structural diversity. Different analytical techniques have been developed for the separation of phenolic compounds in foodstuff. Liquid chromatography (LC) is the technique of choice, employed for the separation of phenolic compounds in functional food matrices [16,17]. Because of the complexity of the matrices, many efforts have been made for the optimization of the analytical LC methodologies to achieve separation. Gas chromatography (GC) is widely used in applications in food analysis, as well. This review gathers all the recent advances concerning the analytical separation of phenolic antioxidant compounds in functional foods within the period 2015-2019.
1.1 Search/Selection Criteria
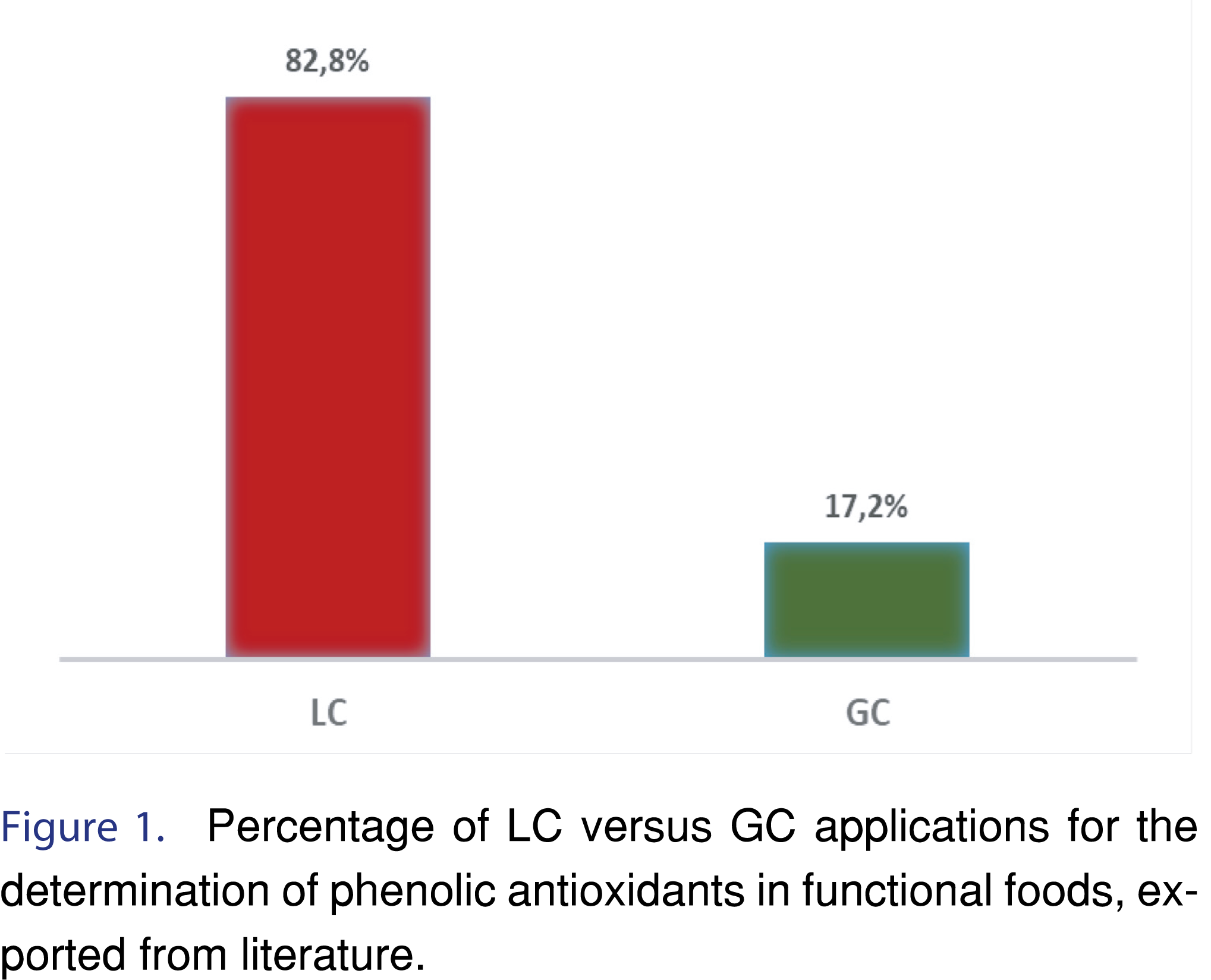
Taking into consideration the emerging need for the development of analytical methodologies that aim at the successful separation of antioxidant compounds, this review summarizes all recent advances in the separation of phenolic antioxidant compounds from food matrices between the years 2015-2019. This time-frame was selected after examining the literature and realizing that Pyrzynska et al. [16] have summarized the analytical developments in the LC separation of phenolic food compounds up to 2015. In the meantime, Capriotti et al. published [18] a review article summarizing all advances in LC and GC methodologies up to 2015. All the LC and GC methodologies that were published after that and have not yet been reviewed, were gathered and discussed in this mini review article. As it is illustrated in Figure 1, the majority of the research papers use LC methodologies for the separation of the phenolic compounds in food matrices, instead of GC. This could be easily explained by the polarity of the phenolic compounds, which need to undergo derivatization before GC analysis, as it is described in the following sections.
2.0 Discussion
2.1. Liquid Chromatography
Low and high molecular weight phenolic compounds can be separated by LC, since it provides a wide number of possible combinations between the stationary and mobile phases [19, 20]. However, conventional chromatographic methods, even if they use High Pressure Liquid Chromatography (HPLC), are considered tedious, time consuming and analysis usually lasts longer than 30 min. Thus, it is essential to imply a target strategy in order to develop rapid and efficient procedures to perform qualitative and quantitative analysis within reduced analysis times. The use of UHPLC gains interest over conventional HPLC, since the chemistry of HPLC columns and dimensions affect resolution and sensitivity [21]. The employment of sub-2 µm particle stationary phases in UHPLC provides narrower peaks, lower detection limits and rapid analysis times [22,23]. Moreover, it decreases the volumes of solvents and waste. The separation that takes place is based on the interactions of the sample with the stationary and mobile phases. Since many different stationary and mobile phase combinations that can occur for the separation of a mixture, the LC modes that can be applied for the separation of phenolic compounds in functional foods are also many, including HPLC and UHPLC in reversed phase (RP) or hydrophilic interaction (HILIC) mode, and multidimensional chromatographic systems, as they are described in the following sections.
2.1.1 High Pressure Liquid Chromatography
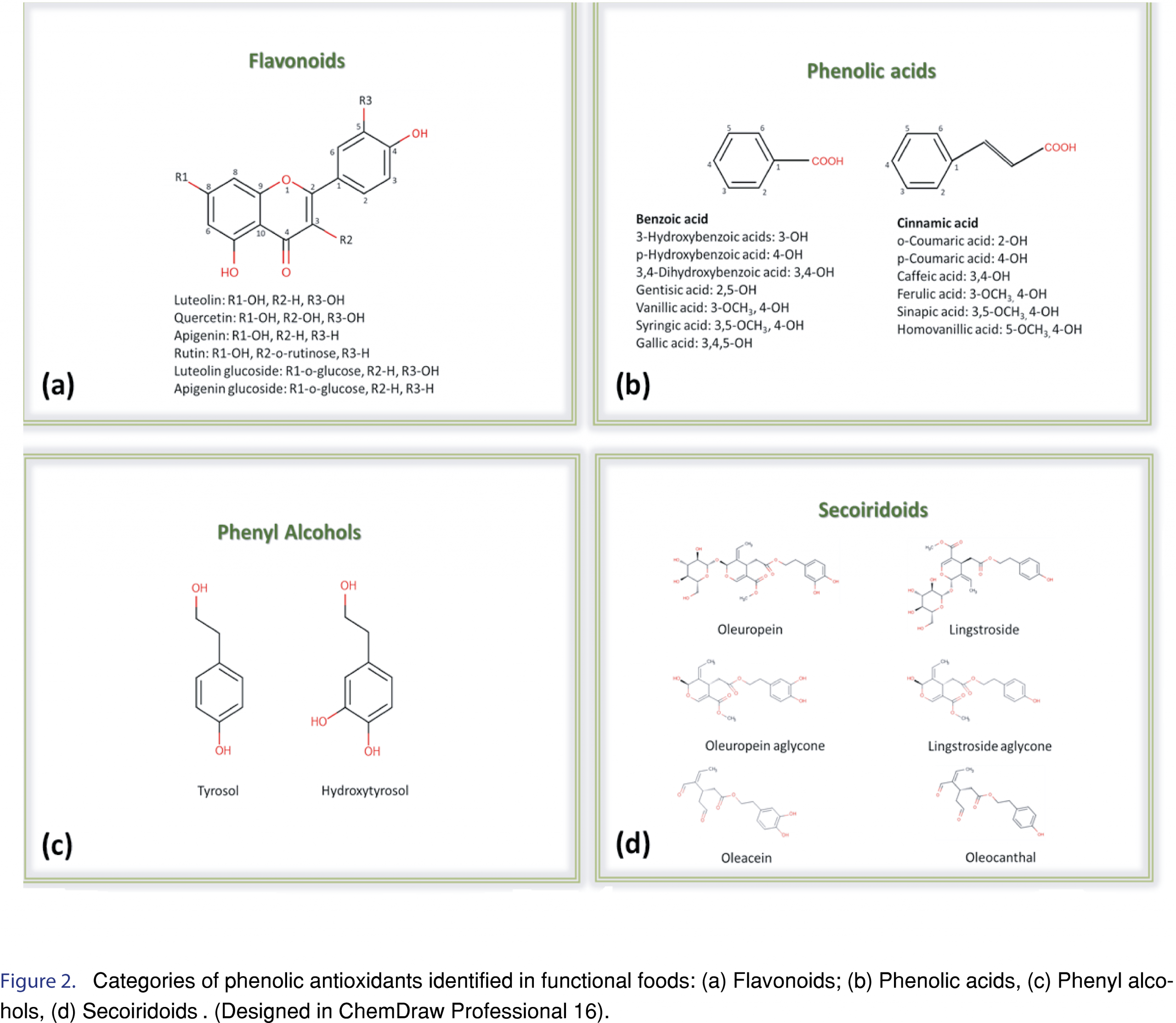
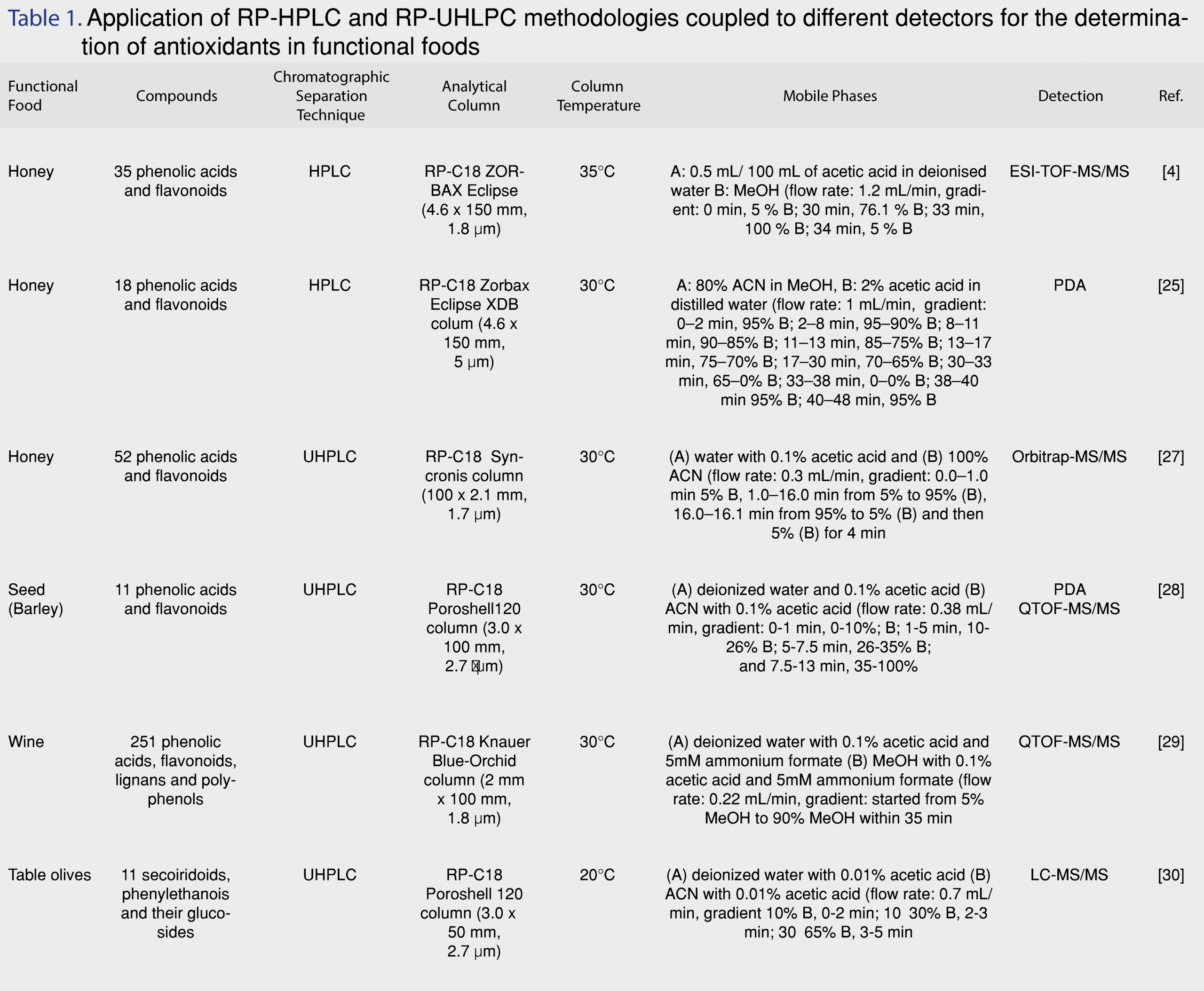
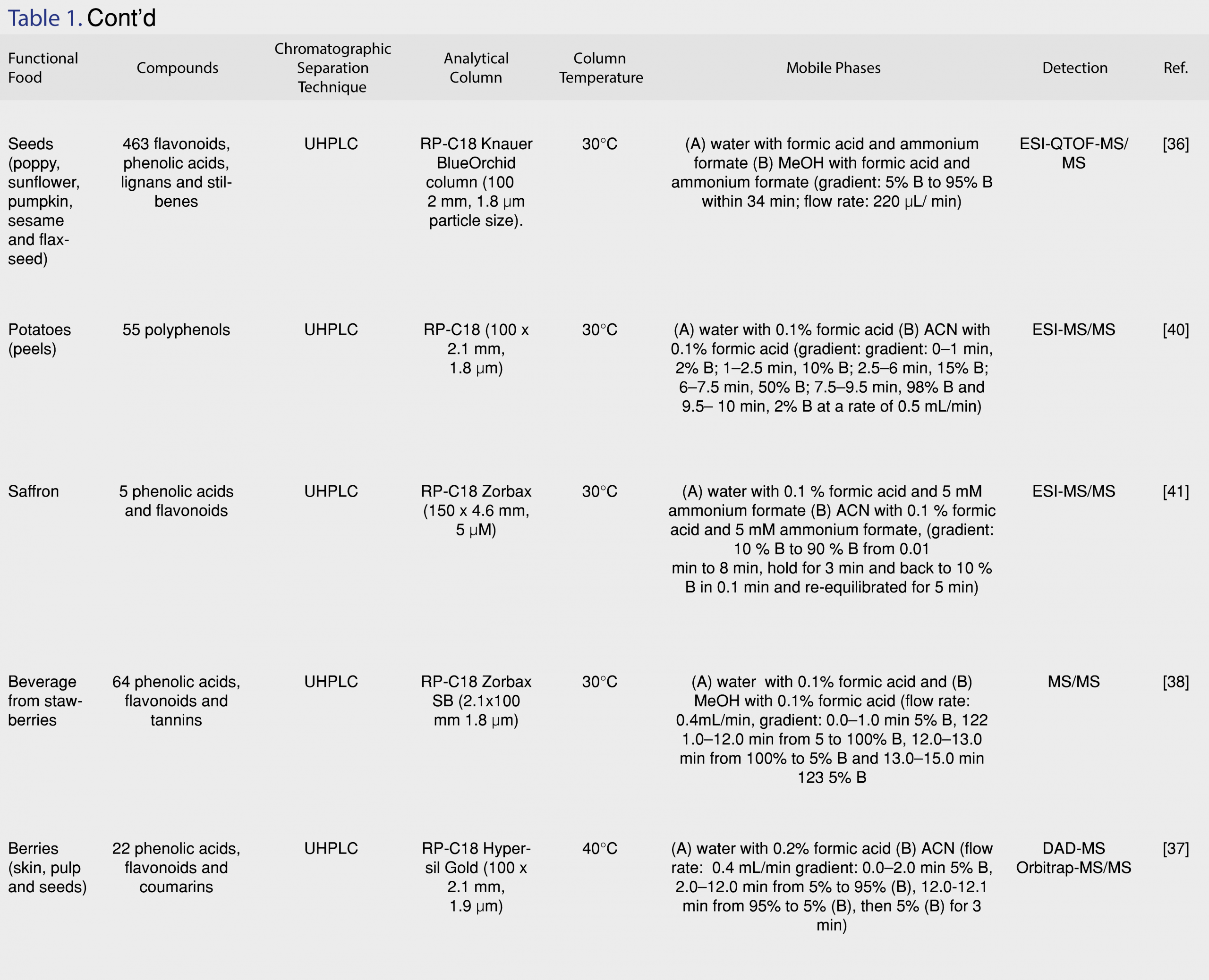
The basic principle of HPLC is that it relies on the different polarities of the constituents of a sample in order to separate them. It is different from standard column chromatography since it requires pressure in order to force the solution pass through the column. HPLC is widely used for the separation of phenolic compounds employing RP-C18 analytical columns. The mobile phase consist of an aqueous solution and an organic solvent, followed by detection with UV, diode array (DAD) or tandem mass spectrometry (MS/MS) [16]. The developments and applications of RP-HPLC methodologies in the separation of phenolic compounds in foods and beverages have been reviewed up to 2014 [16]. Ouchemoukh et al. [24] developed an RP-HPLC-analytical methodology coupled to electrospray time-of-flight mass spectrometry (ESI-TOF-MS) and achieved successful separation for 35 phenolic compounds in honeys, that belonged to different chemical classes, such as phenolic acids, flavonoids and phenyl alcohols (Figure 2) using a Rapid Resolution Liquid Chromatography system (RRLC) equipped with a vacuum degasser, autosampler, binary pump and thermostated column department. They employed an RP-C18 analytical column (4.6 x 150 mm, 1.8 µm particle size, ZORBAX Eclipse plus) and the eluting mobile phase, consisted of A: 0.5 mL/100 mL of acetic acid in deionised water and B: methanol (MeOH), pumped at 1.2 mL/min, applying a multi-step linear gradient, as it is described in Table 1 and the column temperature was maintained at 35°C [24]. Despite the effective separation of the target analytes, the method is time consuming with each analysis run lasting for 40 min. Can et al. [25] developed an HPLC methodology coupled with a UV-Vis detector in 280 and 315 nm, for the determination of 18 phenolic compounds in different type Turkish honeys using an RP Zorbax Eclipse XDB-C18 column (4.6 x 150 mm, 5 µm), thermostated at 30°C and a gradient program with two solvents (A: 80% acetonitrile (ACN) in MeOH, B: 2% acetic acid in distilled water). All targeted compounds were separated with this methodology, but analysis is time consuming. Thus, the major shortcomings of RP-HPLC methodologies are that it requires long elution times and presents reduced selectivity for the separation of co-eluting compounds. Moreover, because of its speed and its reliance on the polarities of the phenolic acids (Figure 2a), flavonoids and their glycosides (Figure 2b), compounds with structural similarity and polarities can possibly exit the chromatographic system nearly at the same or exactly at the same time. Another weakness is adsorption. Since HPLC typically uses glass columns packed with beads of different materials that may bind with the chemicals of the sample mixtures that pass through the column [26]. The increasing need for better chromatographic performance and high throughput resulted in the development of advanced chromatographic techniques, such as UHPLC and HILIC. In addition, multidimensional chromatography shows excellent analytical performance, for the analysis of complex mixtures, increasing peak capacity.
2.1.2 Ultrahigh Pressure Liquid Chromatography
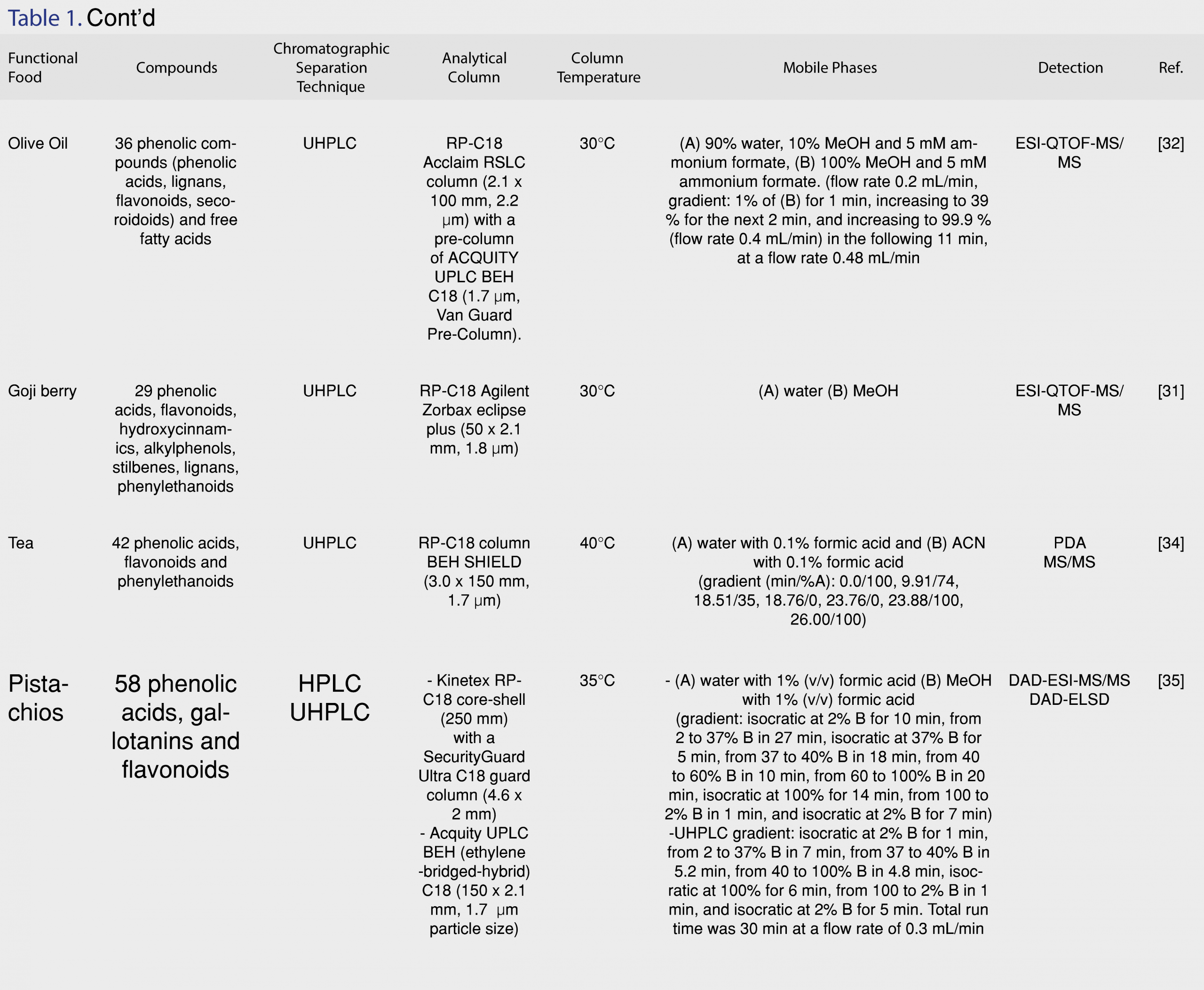
UHPLC is the advanced version of HPLC, using narrow-bore columns that are packed with very small particles (< 2 µm), operating in high pressures. UHPLC systems present high sensitivity, improved resolution, and short time analysis compared to conventional HPLC [6]. Decreasing the size of particles results in gaining higher peak efficiency [21]. Consequently, the sensitivity is higher with narrow peaks and low detection limits (LODs), decreasing the volumes of organic solvents. For the analysis of phenolic constituents in functional foods, RP-C18 columns are frequently used. Vasić et al. [26] performed separation on a Syncronis C18 column (100 x 2.1 mm, 1.7 µm particle size). The mobile phase consisted of (A) water with 0.1% acetic acid and (B) 100% ACN. [27]. An RP-C18 Poroshell120 column (3.0 mm x 100 mm, 2.7 µm) was used for the separation of 11 phenolic acids and flavonoids in barley samples in a UHPLC-PDA and UHPLC-QTOF-MS chromatographic system [28]. UHPLC are applied not only in targeted screening methodologies, but in untargeted metabolomics studies, as well. Rocchetti et al. [28] introduced a metabolomics approach with the development of a UHPLC-ESI-QTOF-MS method for the phenolic profiling of Chardonnay wines from different origins, achieving successful separation and identification for 251 phenolic acids, flavonoids, phenylethanoids, lignans and polyphenols [29]. Johnson et al. [30] used a UHPLC-MS/MS system equipped with an RP-C18 Poroshell 120 column (3.0 x 50 mm, 2.7 µm) for the separation and detection of phenolic alcohols (Figure 2c) and secoiridoids (Figure 2d) in Spanish-style green, Californian-style black ripe, and Greek-style table olives. Hydroxycinnamic acids, flavonols, flavanols, glycoside forms of anthocyanins and flavonols were identified in Italian and Chinese Coji berries with a UHPLC-QTOF-MS with an RP-C18 Agilent Zorbax eclipse plus column (50 x 2.1 mm, 1.8 µm), under a water methanol gradient elution [31]. An RP-UHPLC-ESI-QTOF-MS/MS methodology was developed for the target and non-target determination of phenolic compounds in extra-virgin olive oil samples using an Acclaim RSLC C18 column (2.1 x 100 mm, 2.2 µm) with a pre-column of ACQUITY UPLC BEH C18 (1.7 µm, VanGuard Pre-Column) within 11 min. The separation was operated at 30°C. The solvents used consisted of: (A) 90% H2O, 10% MeOH and 5 mM ammonium formate, (B) 100% MeOH and 5 mM ammonium formate [32]. Thirthy six phenolic compounds were determined through target, suspect and non-target screening, including phenolic acids, flavonoids, lignans and phenylethanoids, while the method also enabled the determination of free fatty acids with retrospective analysis. The extraction procedure was also optimized, suggesting the mixture of MeOH:water (80:20, v/v) as the most appropriate extraction solvent in olive oil samples [33]. Guerreiro Pereira et al. [34] developed a UHPLC-PDA-MS methodology for the identification of antioxidants in medicinal plants which are used as additives in functional beverages, employing a BEH C18, with ethylene bridged hybrid technology, providing extraordinary levels of efficiency, peak asymmetry and chemical stability. Fourty two phenolic acids, flavonoids and phenylethanoids were determined in different organs of H. italicum subsp. Picardii, including roots, vegetative aerial-organs and flowers that are used for tea making with UHPLC coupled to photodiode array (PDA) and MS [34]. Ersan et al. [34] optimized the extraction procedure, developed a novel UHPLC-DAD coupled to evaporative light scattering detector (ELSD) method for the determination of phenolics in pistachios, and compared it to an HPLC method in terms of separation efficiency and quantitation. According to the results, analyses with UHPLC were 3.7 times faster and the core-shell particle HPLC column allowed superior peak resolution [35]. The detected antioxidants exhibited a wide range of polarity, ranging from high water-soluble gallotannins and flavonoid glycosides to less polar anacardic acids. The potential health benefits of nuts consumption were investigated after the development of a UHPLC-ESI-QTOF-MS methodology that was applied in poppy, sunflower, pumpkin, sesame and flaxseed matrices for the determination of lignans. Based on the results, all matrices proved to be excellent sources of phenolics, displaying high antioxidant capacity and worth receiving increasing attention as functional foods, since in addition to their nutritional benefits (vitamins, amino acids, trace elements, fiber), they also contain lignans and flavonoids [36]. Pantelić et al. [37] evaluated the antioxidant capacity of 13 varieties of berries in Serbia with a UHPLC-ESI-MS/MS methodology. A novel functional beverage made from strawberry by fermentation was proposed by Álvarez-Fernández et al. [38]. This interesting approach describes the transformation of strawberries into a beverage by gluconic and acetic fermentation and the subsequent monitoring of the nonanthocyanin content within 90 days of storage in two different temperatures (27-30°C and 4°C). Dudonné et al. [39] reported for the first time the complete characterization of proanthocyanidins in native Canadian berries, identifying over 60 phenolic compounds in less than 10 min with UHPLC-MS/MS and verifying the contribution of their antioxidant compounds in beneficial health effects. UHPLC coupled to ESI-MS/MS has also been applied for the investigation of the antioxidant profile of potatoes [40], saffron [41] and in the polyphenolic characterization of food supplements containing 36 different vegetables, fruits and berries [42]. Machado et al. [43] optimized the phenolic extraction from residues of blackberries by evaluating the influence of temperature (60, 80 and 100°C) and solvent type (water, acidified water pH=2.5, ethanol and ethanol with water 50% v/v) using pressurized liquid extraction (PLE) and selected ethanol with water (50% v/v) as the ideal solvent at 100 °C. All recent applications of RP-HPLC and RP-UHLPC methodologies for the determination of antioxidants in functional foods are summarized in Table 1.
2.1.3 HILIC Chromatography
HILIC is an alternative to RP-HPLC separations of polar, basic or weak acidic samples. HILIC is characterized as normal phase chromatography, using polar columns in aqueous-organic mobile phases for the separation of the analytes [44]. The separation mechanism of HILIC is opposite to RP systems. It uses hydrophilic stationary phases with RP type eluents. Any polar chromatographic surface, such as simple unbonded silica silanol or diol bonded phases, amide bonded phases, amino or anionic bonded phases, zwitterionic bonded phases and cationic bonded phases, can be used for HILIC separations [45]. As for the mobile phase, typically HILIC uses ACN with a small amount of water [38]. Aprotic solvents miscible with water such as THF or dioxane can be used, as well. In order to control the pH and ion strength of the mobile phase, ammonium acetate and ammonium formate are usually added. Other salts like sodium perchlorate can be used in order to increase the mobile phase polarity and effect elution. HILIC has been used for the separation of small phenolic compounds in functional foods, counting fewer applications compared to UHPLC [45]. The selectivity and the retention of phenols is affected by the pH, the fraction of the organic solvent and the concentration of the used buffer. pH affects the retention by shifting the ionization of the column material and the analytes. The ionized analytes exhibit stronger strong retention on HILIC columns, since they are more hydrophilic in their ionized form compared to neutral. Recently, the use of amide-embedded stationary phases for the LC separations of small phenolic compounds has become very popular. Aral et al. [46] developed an amide-embedded silica based stationary phase and studied the retention time behaviors of six different groups of phenolic compounds, including anilines, sudan dyes. The chromatographic behavior of the developed stationary phase was compared to commercially available RP column ACE C-18 under HILIC conditions. According to the experimental data, the functional groups on the stationary phases influence the selectivity probably through secondary interactions with the model compounds. The retentions on the stationary phase of the moderately polar compounds are higher than those of the highly polar compounds, because of the hydrophobic and hydrophilic interactions between the analytes and the stationary phase.
2.1.4 Multidimensional Chromatography
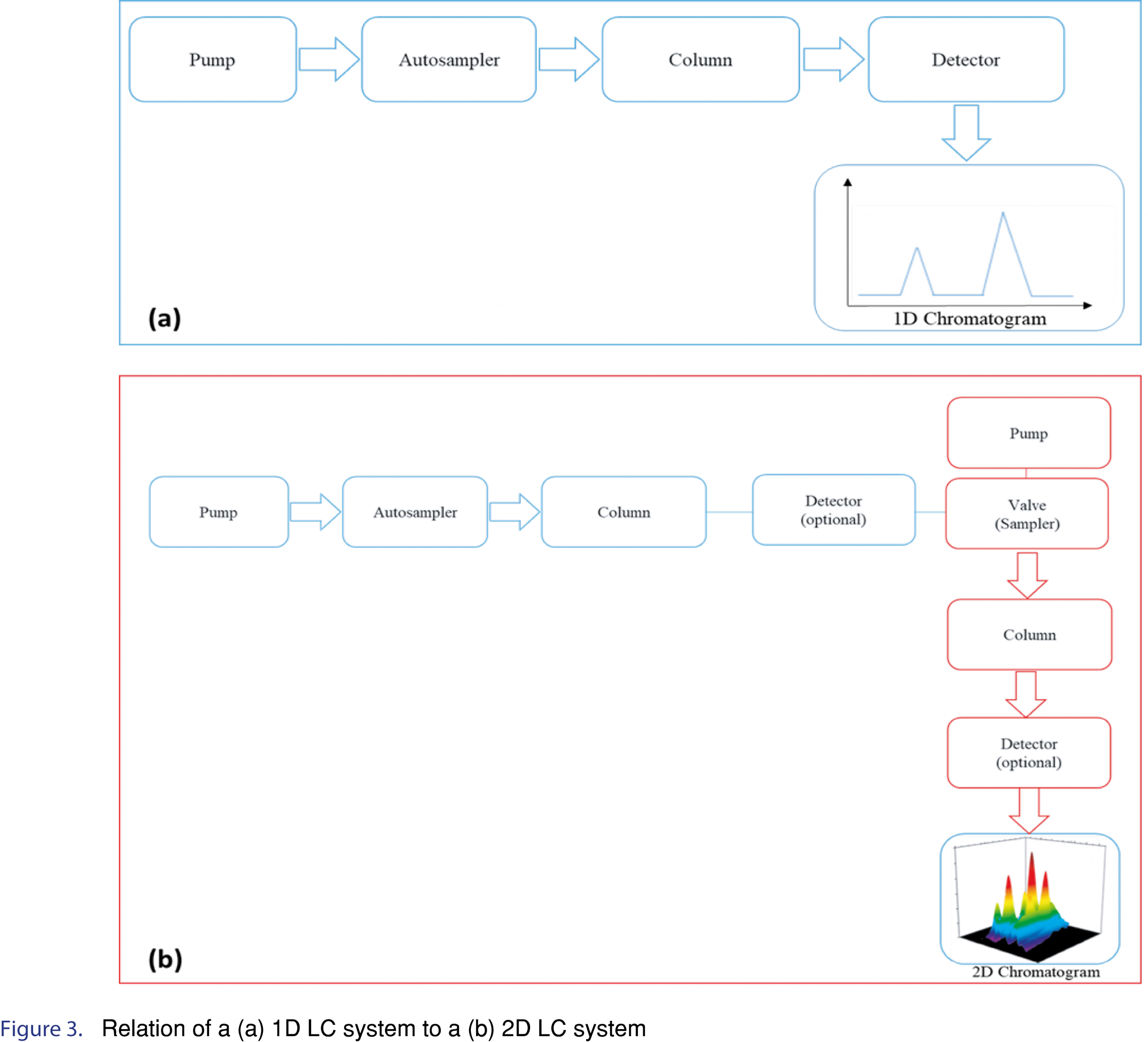
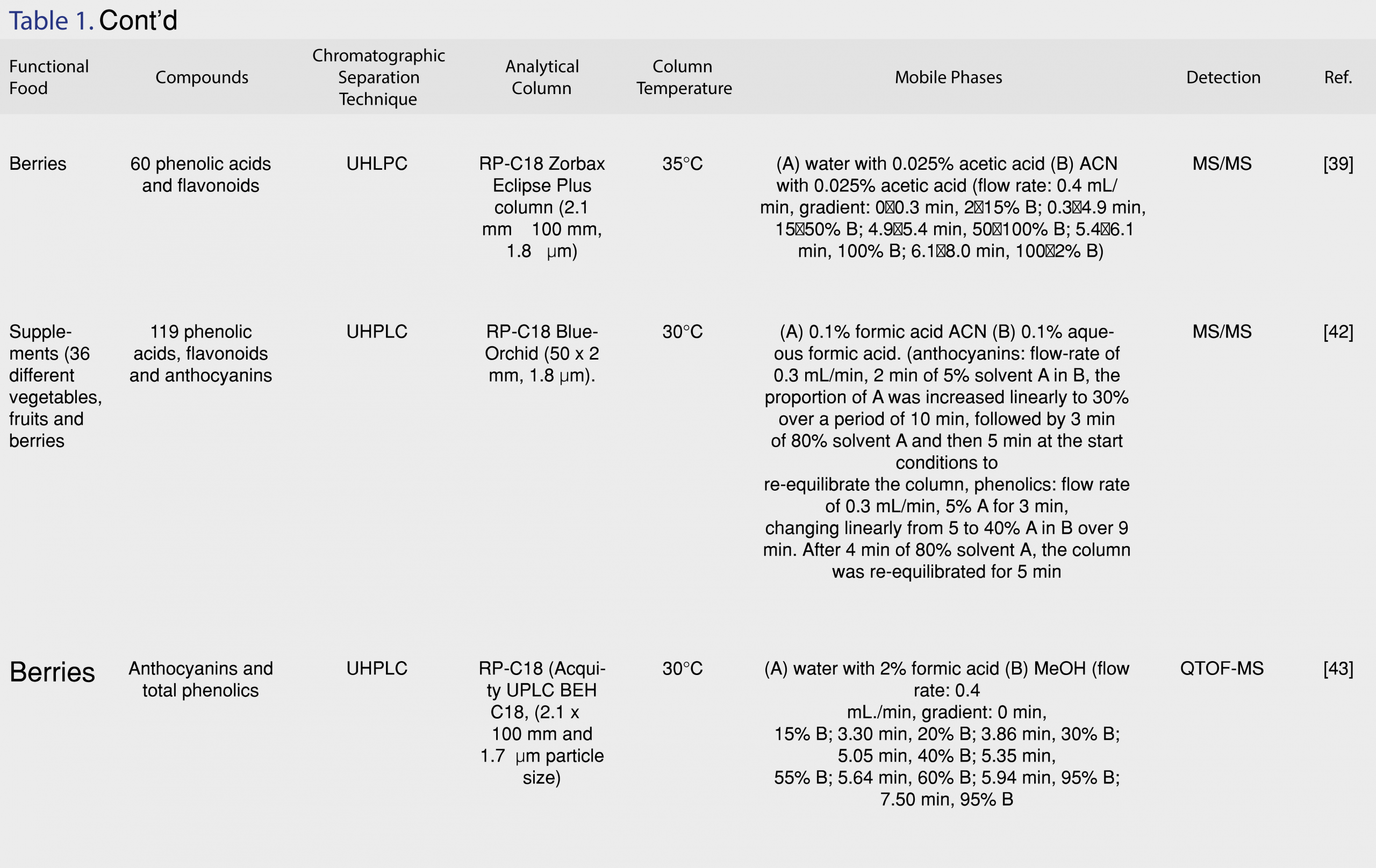
Taking into consideration that food matrices are complex, in some cases a single column chromatographic methodology may not achieve the separation of the antioxidants of interest, especially if the target compounds are isomers or have structural similarity. Comprehensive two-dimensional liquid chromatography (LC x LC) is a promising technique for the analytical separation of phenolics from food matrices in off-line, on-line or stop-flow modes [38]. Two dimensional LC includes one-dimensional (1-D) isocratic and gradient elution LC. Multidimensional systems offer great improvements in the resolving power over 1D-LC. Figure 3 shows the relation between 1D-LC and 2D-LC systems.
Especially, the combination of HILIC and RP is challenging because of the orthogonal group-type separations that are achieved [47]. The on-line hyphenation of HILIC and RP is complicated as a result of the strength of the elution of the mobile phases in both dimensions [48]. To solve this problem, Muller et al. [49] developed a generic HILIC x RP-LC-DAD-MS methodology, utilizing dilution of the first dimension flow and employing large volume injection in the second dimension by kinetic optimization of experimental parameters in an attempt to provide maximum performance [49]. With this methodology, 149 phenolics were tentatively identified in rooibos tea, wine and grapes. In first dimension, separations were performed on an Xbridge Amide column (150 x 1.0 mm, 1.7 µm, Waters) and the mobile phase consisted of (A) 0.1% formic acid in ACN and (B) 0.1% formic acid in water at 11 µL/min flow rate. In second dimension, the first dimension eluent was diluted with 0.1% formic acid in water at 99 µL/min flow rate and a Kinetex C18 column (50 x 3.0 mm, 1.7 µm, Phenomenex, Torrance, USA) was used for the analysis of grape seeds, and a Zorbax Eclipse Plus C18 column (50 x 3.0 mm, 1.8 µm, Agilent) was used for tea, wine and grape analysis. The columns were thermostated at 50°C [49]. Lu et al. [50] developed an online HPLC methodology with silica capillary tube with polyamide coating, coupled to ion mobility spectrometry (IMS) for the determination of nine phenolic acids in seedling roots. The separation was performed by gradient elution: (A) MeOH with 0.1% acetic acid (v/v) on a narrow diameter RP-C18 column with ammonia as the sheath fluid. IMS was employed to assist in the determination of the co-eluted compounds that could not be separated by HPLC, in order to achieve a highly selective and versatile two-dimensional chromatographic analysis. The proposed method enabled effective separation of coumaric acid, chlorogenic acid, protocatechuic acid, caffeic acid, vanillic acid, sinapic acid, ferulic acid, benzoic acid and salicylic acid. The limit of detection (LOD) ranged from 0.05 to 5 ng and the recovery of the spiked samples ranged from 85.10% to 100.51% [50]. In another work, a novel (LC x×LC) method was developed and introduced as a powerful alternative to conventional LC, coupled to DAD and MS for the determination of the polyphenolic fraction of pistachio kernels from different geographical origins. Pistachios are a source of different groups of valuable phytochemicals such as flavan-3-ols, anthocyanins, proanthocyanidins, flavonoids, phenolic acids and stilbenes, presenting excellent biological activities. A 150-mm micro-bore cyano column (2.7 µm) and 50-mm superficially porous C18 silica column (2.7 µm) were employed in the first and second dimension, respectively, to overcome conventional LC limitations, especially in terms of resolving power [51]. Lu et al. developed an LC×LC-ESI-MS/MS method with shifted gradients in the second dimension for the phenolic characterization of pomegranates, providing simultaneous separation and identification for 37 phenolic bioactive constituents [52]. Toro-Uribe et al. [53] evaluated the phenolic content of cocoa with the determination of 30 main compounds, including 3 xanthines, 2 flavan-3-ols and 24 oligomeric procyanidins, using a novel on-line LC x LC-MS/MS methodology, after studying different modulation strategies to enhance the method performance. High resolving power and peak capacity were achieved with focusing modulation set-up. The use of active modulation with the addition of a make-up flow increased the solvent strength mismatch produced between dimensions. Recently, an innovative 3-dimensional LC x LC-IMS methodology was introduced by Venter et al. [54] for the analysis of phenolic compounds in various natural products. The combination of HILIC, RP-LC and IMS provided excellent separation of the phenolic compounds in three dimensions. The incorporation of IMS improved MS sensitivity and mass spectral data quality. IMS also enabled the separation of trimeric procyanidin isomeric species, which could not be differentiated by High Resolution Mass Spectrometry (HR-MS) or HILIC x RP-LC. A comprehensive LC x LC method was developed coupled to DAD and MS to obtain the phenolic profiles of grapevine canes from several varieties [55]. The method was optimized and the combination of diol and C18 columns produced the best results, enabling the determination of 81 phenolic components in around 80 min, showing good separation capability, effective peak capacity and orthogonality. Table 2 presents all recent applications of LC x LC methodologies for the determination of antioxidants in functional foods.
Figures and Tables
[Click to enlarge]
2.2 Gas Chromatography
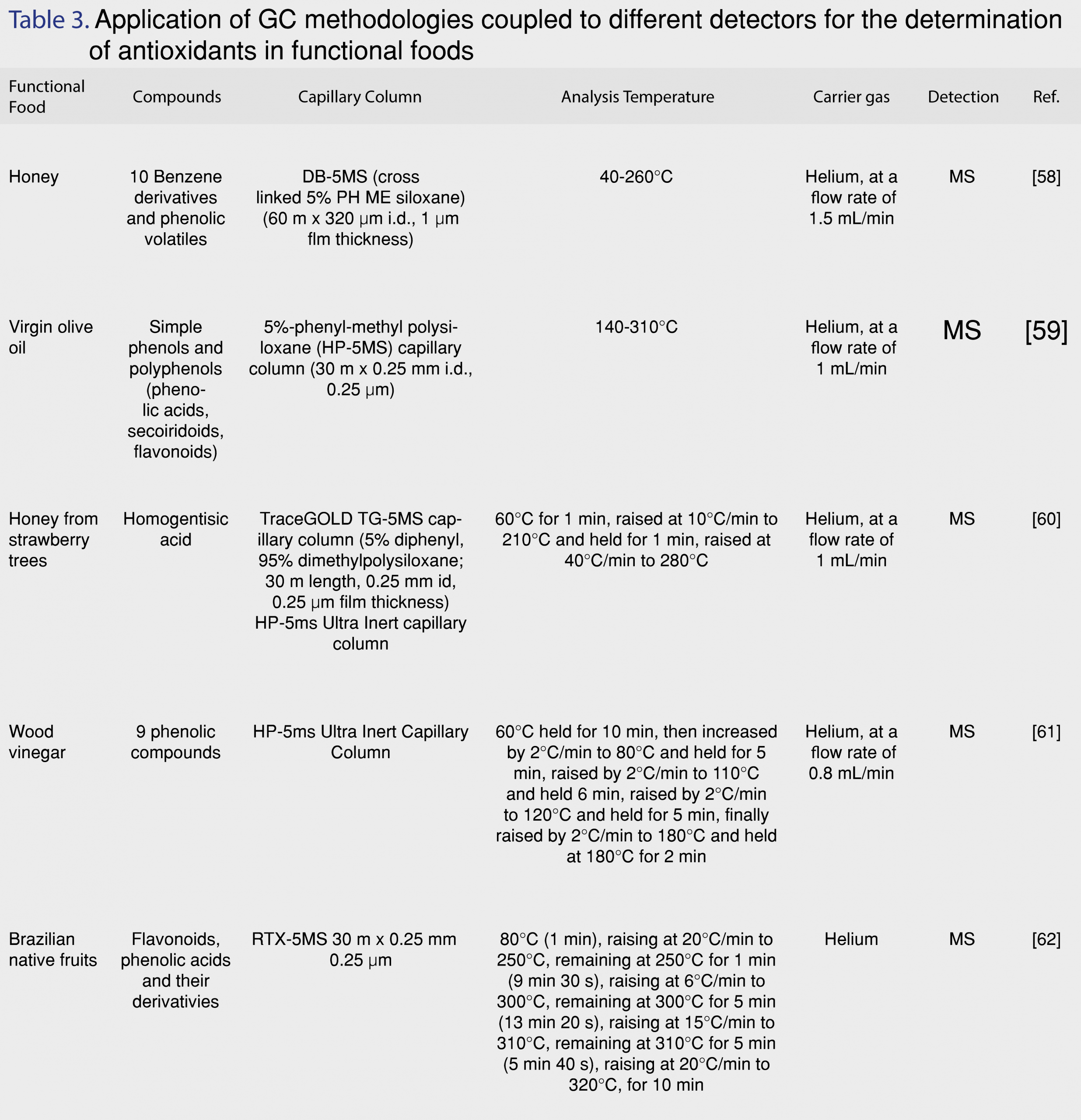
The application of GC is vital for the analysis of non-polar and semi-polar phenolic antioxidants. Despite the fact that GC and LC are complementary techniques, the evolution of HPLC led both analysts and instrument manufacturers to use primarily HPLC [56]. GC is mainly applied for the analysis of the lipid fraction of functional foods. There are only a few works using GC-MS for the identification of phenolic antioxidants in functional foods. An interesting study by da Silva et al. describes the application of GC in the determination of phenolic compounds in native fruits from South America [57]. Karabagias et al. [58] proposed the application of a non-target headspace solid-phase microextraction (HS-SPME) methodology in combination with GC-MS for the determination of volatiles in greek honeys and the exploitation of natural organic compounds. In another interesting study, a GC-MS method was developed complimentary to an LC-MS methodology for the simultaneous determination of more than 40 minor compounds that belonged to different chemical classes, just in a single run, in order to evaluate the profiles of different virgin olive oils that were produced in laboratory scale [59]. Because of the chemical complexicity of the compounds, the chromatographic conditions were optimized to cover a wide range of volatilities. Thus, the temperature ramp was tested from 120°C to 320°C at 3°C/min. Finally, 4 °C/min ramp from 140°C to 310°C produced chromatograms with the best resolution/analysis time ratio. The injection volume was also optimized, testing injections of 0.1, 0.5 and 1 µL of sample in splitless and split modes (1:10, 1:25 1:50 and 1:75 as split ratios) and 1 µL was selected as the optimum injection volume with a split of 1:25 [59]. A GC-MS method was developed and validated for the determination of homogentisic acid (HGA) in strawberry tree honey [60]. The method was optimized and ethyl acetate was selected as the most effective solvent for the extraction, among dichloromethane and ethyl ether. HGA recovery improved with the addition of NaCl and a decrease in pH, since the addition of salt influenced the ionic strength of the solution and decreased the solubility of HGA in the aqueous phase, enhancing its partitioning in the organic [60]. In another study, the volatile antioxidant profile of wood vinegar was evaluated using GC-MS. A total of 17 chemical compounds were determined and 9 of them were phenolic compounds (2,6-dimethoxyphenol, 3,5-dimethoxy-4-hydroxytoluene, and 1-(4-hydroxy-3,5-dimethoxyphenyl)-ethanone, 2-methoxyphenol, 4-ethyl-2-methoxyphenol, and 1-(4-hydroxy-3-methoxyphenyl)-2-propanone, catechol, 3-methoxy-1,2-benzenediol, and 4-methyl-1,2,-benzenediol) [61]. In this study, more than 70% of the chemical components were identified as phenolic compounds and the the antioxidant activity assay showed great antioxidant activity [61]. GC-MS was employed for the determination of cinnamic acid, protocatechuic acid, p-coumaric acid, m-coumaric acid, gallic acid, sinapic acid, 6,7-dihydroxycoumarin, β-D-glycopyranoside, epicatechin, kaempferol, quercetin and myricetin in the ethanolic axtracts of leaves, seeds and pulp of 4 Brazilian native fruit species [62]. Prior to GC-MS analysis, derivatization was carried out with MSTFA in order to reduce the polarity of the functional groups and facilitate separation. Gallic acid and epicatechin were the most abundant antioxidants in these Brazilian fruits [62]. All applications of GC-MS in the determination of antioxidants in functional foods are summarized in Table 3.
2.3 Future Perspectives
The major goals in analytical separation techniques will always be the same: increase accuracy and selectivity, decrease detection and quantification limits, solvents volume and costs. Increasing analysis speed will always be vital, as well. The automation of the sample preparation process and the use of microextraction techniques [62] in combination with novel hybrid on-line systems [63] could make analytical operations more robust and precise, opening new areas of research. LC and GC conditions should be developed and optimized to provide short time of analysis, while providing the adequate selectivity and accuracy. The automation of the extraction process and the use of two-dimensional chromatographic systems, coupled to high resolution detection, despite the instrumentation cost, will play a major role in the future investigation of complex food matrices.
3.0 Conclusions
The aim of this review was to gather all the recent advances in the development and optimization of LC methodologies for the separation of phenolic compounds in functional foods. The use of HPLC provides increased throughput. The application of UHPLC offers several benefits, compared to conventional HPLC, such as better separation efficiency and sensitivity, decreasing solvents volumes and waste and shortening the time of analysis. In addition, the development of comprehensive multidimensional LC systems proves promising, offering high resolution, and has already found several applications in the investigation of the phenolic profile, especially in complex matrices, like foodstuff, and is expected to play a significant role in the field of analytical separations in the future.
4.0 Summary Points list
- Functional foods gain considerable attention because of their high nutritional value and antioxidant activity.
- UHPLC in reversed phase RP is widely used for the separation of phenolic compounds in food matrices.
- The development of multidimensional methods is advantageous compared to conventional HPLC methodologies for phenolic analysis of complex matrices.
5.0 References
1. Mark R, Lyu X, Lee JJL, Parra-Saldívar R, Chen WN. Sustainable production of natural phenolics for functional food applications. J Funct Foods 57, 233-254 (2019). [CrossRef]
2. Valduga AT, Gonçalves IL, Magri E, Delalibera Finzer JR. Chemistry, pharmacology and new trends in traditional functional and medicinal beverages. Food Res Int 120, 478-503 (2019). [CrossRef]
3. Battino M, Forbes-Hernández TY, Gasparrini M, Afrin S, Cianciosi D, Zhang J, et al. Relevance of functional foods in the Mediterranean diet: the role of olive oil, berries and honey in the prevention of cancer and cardiovascular diseases. Crit Rev Food Sci Nutr 59(6), 893-920 (2019). [CrossRef]
4. Nazir M, Arif S, Khan RS, Nazir W, Khalid N, Maqsoodd S. Opportunities and Challenges for Functional and Medicinal Beverages: Current and Future Trends. Trends Food Sci 88, 513-526 (2019). [CrossRef]
5. Sharma A, Kaur M, Katnoria JK, Nagpal AK. Polyphenols in food: Cancer prevention and apoptosis induction. Curr Med Chem 25(36), 4740-4757 (2018). [CrossRef]
6. Fabroni S, Ballistreri G, Amenta M, Romeo FV, Rapisarda P. Screening of the anthocyanin profile and in vitro pancreatic lipase inhibition by anthocyanin-containing extracts of fruits, vegetables, legumes and cereals. J Sci Food Agric 96(14), 4713-4723 (2016). [CrossRef]
7. Lutz M, Fuentes E, Ávila F, Alarcón M, Palomo I. Roles of phenolic compounds in the reduction of risk factors of cardiovascular diseases Molecules 366, (2019). [CrossRef]
8. Cianciosi D, Forbes-Hernández TY, Author E, Afrin S, Gasparrini M, Reboredo-Rodriguez P, et al. Phenolic compounds in honey and their associated health benefits: A review. Molecules 23(9), (2018). [CrossRef]
9. Brewer MS. Natural Antioxidants: Sources, Compounds, Mechanisms of Action, and Potential Applications. Compr Rev Food Sci F 10, 221-248 (2011). [CrossRef]
10. Sherkatolabbasieh HR, Hasanvand A, Abbaszadeh S, Farzan B. Effect of natural antioxidants and important herbal medicines on blood infections and sepsis: A systematic review. Int J Pharma Sci 10(2), 1372-1381 (2019). [CrossRef]
11. Xu D, Hu M-J, Wang Y-Q, Cui Y-L. Antioxidant activities of quercetin and its complexes for medicinal application. Molecules. (2019). [CrossRef]
12. Jurikova T, Skrovankova S, Mlcek J, Balla S, Snopek L. Bioactive compounds, antioxidant activity, and biological effects of European cranberry (vaccinium oxycoccos). Molecules. 24(1), (2019). [CrossRef]
13. do Carmo MAV, Pressete CG, Marques MJ, Granato D, Azevedo L. Polyphenols as potential antiproliferative agents: scientific trends. Curr Opin Food Sci 24, 26-35 (2018). [CrossRef]
14. Tresserra-Rimbau A, Lamuela-Raventos RM, Moreno JJ. Polyphenols, food and pharma. Current knowledge and directions for future research. Biochem Pharmacol 156, 186-195 (2018). [CrossRef]
15. Mark R, Lyu X, Lee JJL, Parra-Saldívar R, Chen WN. Sustainable production of natural phenolics for functional food applications. J Funct Foods 57, 233-254 (2019). [CrossRef]
16. Pyrzynska K, Sentkowska A. Recent Developments in the HPLC Separation of Phenolic Food Compounds. Crit Rev Anal Chem 45(45), 41-51 (2015). [CrossRef]
17. Cheiran KP, Raimundo VP, Manfroi V, Anzanello MJ, Kahmann A, Rodrigues E, et al. Simultaneous identification of low-molecular weight phenolic and nitrogen compounds in craft beers by HPLC-ESI-MS/MS. Food Chem 286(15), 113-122 (2019). [CrossRef]
18. Capriotti AL, Cavaliere C, Foglia P, Piovesana S, Ventura S. Chromatographic methods coupled to mass spectrometry detection for the determination of phenolic acids in plants and fruits. J Liq Chromatogr Relat Technol 38(3), 353-370 (2015). [CrossRef]
19. Esteki M, Shahsavari Z, Simal-Gandara J. Food identification by high performance liquid chromatography fingerprinting and mathematical processing. Food Res Int 122, 303-317 (2019). [CrossRef]
20. Ganzera M, Sturm S. Recent advances on HPLC/MS in medicinal plant analysis—An update covering 2011–2016. J Pharma Biomed Anal 147, 211-233 (2018). [CrossRef]
21. Fekete S, Schappler J, Veuthey J-L, Guillarme D. Current and future trends in UHPLC. Trends Anal Chem 63, 2-13 (2014). [CrossRef]
22. Dores-Sousa JL, De Vos J, Eeltink S. Resolving power in liquid chromatography: A trade-off between efficiency and analysis time. J Sep Sci 42(1), 35-80 (2019). [CrossRef]
23. Dong MW, Zhang K. Ultra-high-pressure liquid chromatography (UHPLC) in method development. Trends Anal Chem 63, 21-30 (2014). [CrossRef]
24. Ouchemoukh S, Amessis-Ouchemoukh N, Gómez-Romero M, Aboud F, Guiseppe A, Fernández-Gutiérrez A, et al. Characterisation of phenolic compounds in Algerian honeys by RP-HPLC coupled to electrospray time-of-flight mass spectrometry. LWT – Food Sci Technol 85, 460-469 (2017). [CrossRef]
25. Can Z, Yildiz O, Sahin H, Akyuz Turumtay E, Silici S, Kolayli S. An investigation of Turkish honeys: Their physico-chemical properties, antioxidant capacities and phenolic profiles. Food Chem 180, 133-141 (2015). [CrossRef]
26. Kalili KM, A. dV. Recent developments in the HPLC separation of phenolic compounds. J Sep Sci 34, 854-876 (2011). [CrossRef]
27. Vasić V, Gašić U, Stanković D, Lušić D, Vukićlušić D, Milojković-Opsenica D, et al. Towards better quality criteria of European honeydew honey: phenolic profile and antioxidant capacity. Food Chem 274, 629-641 (2018). [CrossRef]
28. Rao S, Santhakumar AB, Chinkwo KA, Blanchard CL. Q-TOF LC/MS identification and UHPLC-Online ABTS antioxidant activity guided mapping of barley polyphenols. Food Chem 15, 323-328 (2018). [CrossRef]
29. Rocchetti G, Gatti M, Bavaresco L, Lucini L. Untargeted metabolomics to investigate the phenolic composition of Chardonnay wines from different origins. J Food Compost Anal 71, 87-93 (2018). [CrossRef]
30. Johnson R, Melliou E, Zweigenbaum J, Mitchell AE. Quantitation of Oleuropein and Related Phenolics in Cured Spanish-Style Green, California-Style Black Ripe, and Greek-Style Natural Fermentation Olives. J Agric Food Chem 66, 2121-2128 (2018). [CrossRef]
31. Rocchetti G, Chiodelli G, Giuberti G, Ghisoni S, Baccolo G, Blasi F, et al. UHPLC-ESI-QTOF-MS profile of polyphenols in Goji berries (Lycium barbarum L.) and its dynamics during in vitro gastrointestinal digestion and fermentation. J Funct Foods 40, 564-572 (2018). [CrossRef]
32. Kalogiouri NP, Alygizakis NA, Aalizadeh R, Thomaidis NS. Olive oil authenticity studies by target and nontarget LC–QTOF-MS combined with advanced chemometric techniques. Anal Bioanal Chem 408 7955-7970 (2016). [CrossRef]
33. Kalogiouri NP, Aalizadeh R, Thomaidis NS. Investigating the organic and conventional production type of olive oil with target and suspect screening by LC-QTOF-MS, a novel semi-quantification method using chemical similarity and advanced chemometrics. Anal Bioanal Chem 409, 5413-5426 (2017). [CrossRef]
34. Guerreiro Pereira C, Barreira L, Bijttebier S, Pieters L, Neves V, Rodrigues MJ, et al. Chemical profiling of infusions and decoctions of Helichrysum italicum subsp. picardii by UHPLC-PDA-MS and in vitro biological activities comparatively with green tea (Camellia sinensis) and rooibos tisane (Aspalathus linearis). J Pharma Biomed Analysis 145, 593-603 (2017). [CrossRef]
35. Erşan S, Güçlü Üstündağ Ö, Carle R, Schweiggert RM. Determination of pistachio (Pistacia vera L.) hull (exo- and mesocarp) phenolics by HPLC-DAD-ESI/MSn and UHPLC-DAD-ELSD after ultrasound-assisted extraction. J Food Compost Analysis 103-114 (2017). [CrossRef]
36. Ghisoni S, Chiodelli G, Rocchetti G, Kane D, Lucini L. UHPLC-ESI-QTOF-MS screening of lignans and other phenolics in dry seeds for human consumption. J Funct Foods 34, 229-236 (2017). [CrossRef]
37. Pantelić MM, Dabić Zagorac DČ, Davidović SM, Todić SR, Bešlić ZS, Gašić UM, et al. Identification and quantification of phenolic compounds in berry skin, pulp, and seeds in 13 grapevine varieties grown in Serbia. Food Chem 15, 243-252 (2016). [CrossRef]
38. Álvarez-Fernández MA, Hornedo-Ortega R, Cerezo AB, Troncoso AM, García-Parrilla MC. Determination of Nonanthocyanin Phenolic Compounds Using High-Resolution Mass Spectrometry (UHPLC-Orbitrap MS/MS) and Impact of Storage Conditions in a Beverage Made from Strawberry by Fermentation. J Agric Food Chem 64(6), 1367-1376 (2016). [CrossRef]
39. Dudonné S, Dubé P, Anhê FF, Pilon G, Marette A, Lemire M, et al. Comprehensive analysis of phenolic compounds and abscisic acid profiles of twelve native Canadian berries. Journal Food Compost Anal 44, 214-224 (2015). [CrossRef]
40. Kumari B, Tiwari BK, Hossain MB, Rai DK, Brunton NP. Ultrasound-assisted extraction of polyphenols from potato peels: profiling and kinetic modelling. Int J Food Sci Technol 52(6), 1432-1439 (2017). [CrossRef]
41. Anuar N, Taha RM, Mahmad N, Mohajer S, Musa SANC, Abidin ZHZ. Correlation of colour, antioxidant capacity and phytochemical diversity of imported saffron by principal components analysis. Pigm Resin Technol 46(2), 107-114 (2017). [CrossRef]
42. Bresciani L, Calani L, Cossu M, Mena P, Sayegh M, Ray S, et al. (Poly)phenolic characterization of three food supplements containing 36 different fruits, vegetables and berries. Pharma Nutrition 3(2), 11-19 (2015). [CrossRef]
43. Machado APDF, Pasquel-Reátegui JL, Barbero GF, Martínez J. Pressurized liquid extraction of bioactive compounds from blackberry (Rubus fruticosus L.) residues: A comparison with conventional methods. Food Res Int 77, 675-683 (2015). [CrossRef]
44. Qiao L, Shi X, Xu G. Recent advances in development and characterization of stationary phases for hydrophilic interaction chromatography. Trends Anal Chem 81, 22-33 (2016). [CrossRef]
45. Bernal J, Ares AM, Pól J, Wiedmer SK. Hydrophilic interaction liquid chromatography in food analysis. J Chromatogr A 1218(42), 7438-7452 (2011). [CrossRef]
46. Aral H, Aral T, Çelik KS, Ziyadanogulları B, Ziyadanogulları R. HPLC Separation of Different Groups of Small Polar Compounds on a Novel Amide‑Embedded Stationary Phase. Chromatographia 77, 771-781 (2014). [CrossRef]
47. De Villiers A, Venter P, Pasch H. Recent advances and trends in the liquid-chromatography-mass spectrometry analysis of flavonoids. J Chromatogr A 1430, 16-78 (2015). [CrossRef]
48. Malerod H, Lundanes E, Greibrokk T. Recent advances in on-line multidimensional liquid chromatography. Anal Methods 2(2), 110-122 (2010). [CrossRef]
49. Muller M, Tredoux AGJ, de Villiers A. Application of Kinetically Optimised Online HILIC × RP-LC Methods Hyphenated to High Resolution MS for the Analysis of Natural Phenolics. Chromatographia 17(1), 181-196 (2019). [CrossRef]
50. Lu Y, Guo J, Yu J, Guo J, Jia X, Liu W, et al. Two-dimensional analysis of phenolic acids in seedling roots by high performance liquid chromatography-electrospray ionization-ion mobility spectrometry. Anal Methods 11(5), 610-617 (2018). [CrossRef]
51. Arena K, Cacciola F, Mangraviti D, Zoccali M, Rigano F, Marino N, et al. Determination of the polyphenolic fraction of Pistacia vera L. kernel extracts by comprehensive two-dimensional liquid chromatography coupled to mass spectrometry detection. Anal Bioanal Chem 411(19), 4819-4829 (2019). [CrossRef]
52. Russo M, Cacciola F, Arena K, Mangraviti D, de Gara L, Dugo P, et al. Characterization of the polyphenolic fraction of pomegranate samples by comprehensive two-dimensional liquid chromatography coupled to mass spectrometry detection. Nat Prod Res 1-7, (2019). [CrossRef]
53. Toro-Uribe S, Montero L, López-Giraldo L, Ibáñez E, Herrero M. Characterization of secondary metabolites from green cocoa beans using focusing-modulated comprehensive two-dimensional liquid chromatography coupled to tandem mass spectrometry. Anal Chim Acta 1036, 204-213 (2018). [CrossRef]
54. Venter P, Muller M, Vestner J, Stander MA, Tredoux AGJ, Pasch H, et al. Comprehensive Three-Dimensional LC × LC × Ion Mobility Spectrometry Separation Combined with High-Resolution MS for the Analysis of Complex Samples. Anal Chem 90(19), 11643-11650 (2018). [CrossRef]
55. Montero L, Sáez V, von Baer D, Cifuentes A, Herrero M. Profiling of Vitis vinifera L. canes (poly)phenolic compounds using comprehensive two-dimensional liquid chromatography. J Chromatogr A 1536(9), 205-215 (2018). [CrossRef]
56. Lehotay SJ, Hajslova J. Application of gas chromatography in food analysis. Trends Anal Chem 21, 686-697 (2002). [CrossRef]
57. da Silva APG, Spricigo PC, Purgatto E, de Alencar SM, Jacomino AP. Volatile Compounds Determined by SPME-GC, Bioactive Compounds, In Vitro Antioxidant Capacity and Physicochemical Characteristics of Four Native Fruits from South America. Plant Foods Hum Nutr (2019). DOI: 10.1007/s11130-019-00745-7. [CrossRef]
58. Karabagias K, Nikolaou C, Karabagias V. Volatile fngerprints of common and rare honeys produced in Greece: in search of PHVMs with implementation of the honey code. Eur Food Res Technol 245, 2 (2018). [CrossRef]
59. Olmo-García L, Polari JJ, Li X, Bajoub A, Fernández Gutiérrez A, Wang SC, et al. Deep insight into the minor fraction of virgin olive oil by using LC-MS and GCMS multi-class methodologies. Food Chem 261, 184-193 (2018). [CrossRef]
60. Brcic-KaraconjI I, Jurica K. Development and Validation of a GC-MS Method for the Analysis of Homogentisic Acid in Strawberry Tree (Arbutus unedo L.) Honey J AOAC Int 100(4), 889-892 (2017). [CrossRef]
61. Yang YF, Yang C-H, Liang M-T, Gao ZJ, Wu YW, Chuang L-Y. Chemical Composition, Antioxidant, and Antibacterial Activity of Wood Vinegar from Litchi chinensis. Molecules 21(1150), 1-10 (2016). [CrossRef]
62. Infante J, Rosalen PL, Lazarini JG, Franchin M, Alencar SM. Antioxidant and Anti-Inflammatory Activities of Unexplored Brazilian Native Fruits. PLoS One 11(4), (2016). [CrossRef]
63. Kalogiouri, NP, Anthemidis AN, Advances in On-Line Hydride Generation Atomic Spectrometric Determination of Arsenic. Anal Lett 46(11), 1672-1704 (2013). [CrossRef]
64. Kalogiouri, NP, Samanidou, VF, Recent Trends in the Development of Green Microextraction Techniques for the Determination of Hazardous Organic Compounds in Wine. Curr Anal Chem (2019). [CrossRef]
All site content, except where otherwise noted, is licensed under a Creative Commons Attribution 4.0 License